Working Hours
Mon - Sat 9:00 - 6:00TEICONOVA 400 MG
Price 400 INR/ Vial
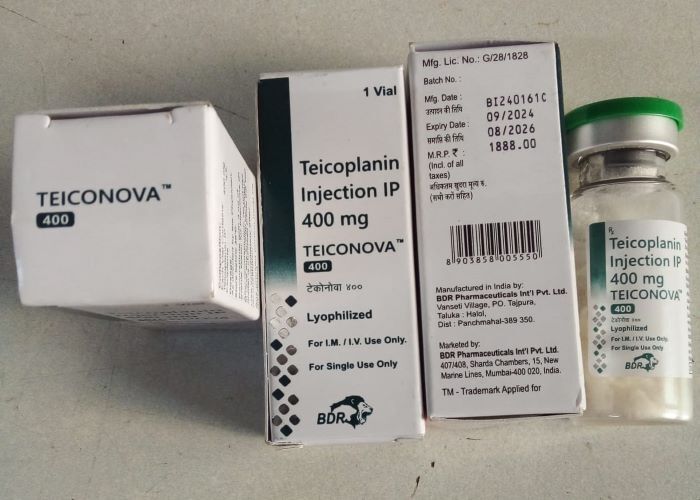
TEICONOVA 400 MG Specification
- Drug Type
- Injection
- Physical Form
- Liquid
- Recommended For
- Doctor
- Dosage Guidelines
- As per Instructions
- Storage Instructions
- Cool & Dry Place
- Molecule
- Teicoplanin Injection
- Strength
- 40mg
TEICONOVA 400 MG Trade Information
- Minimum Order Quantity
- 50 Vials
- Supply Ability
- 1000 Vials Per Month
- Delivery Time
- 7 Days
About TEICONOVA 400 MG
BRAND : TEICONOVA 400 MGCONTENT : TEICOPLANIN INJECTION 400 MG
COMPANY : BDR PHARMACEUTICALS


Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in IV Fluids Category
FOSFOFAST POWDER 3 GM SACHET
Price 269.0 INR / Piece
Minimum Order Quantity : 20 Pieces
Dosage Guidelines : As per Instructions
Physical Form : Liquid
Drug Type : Injection
Storage Instructions : Cool & Dry Place
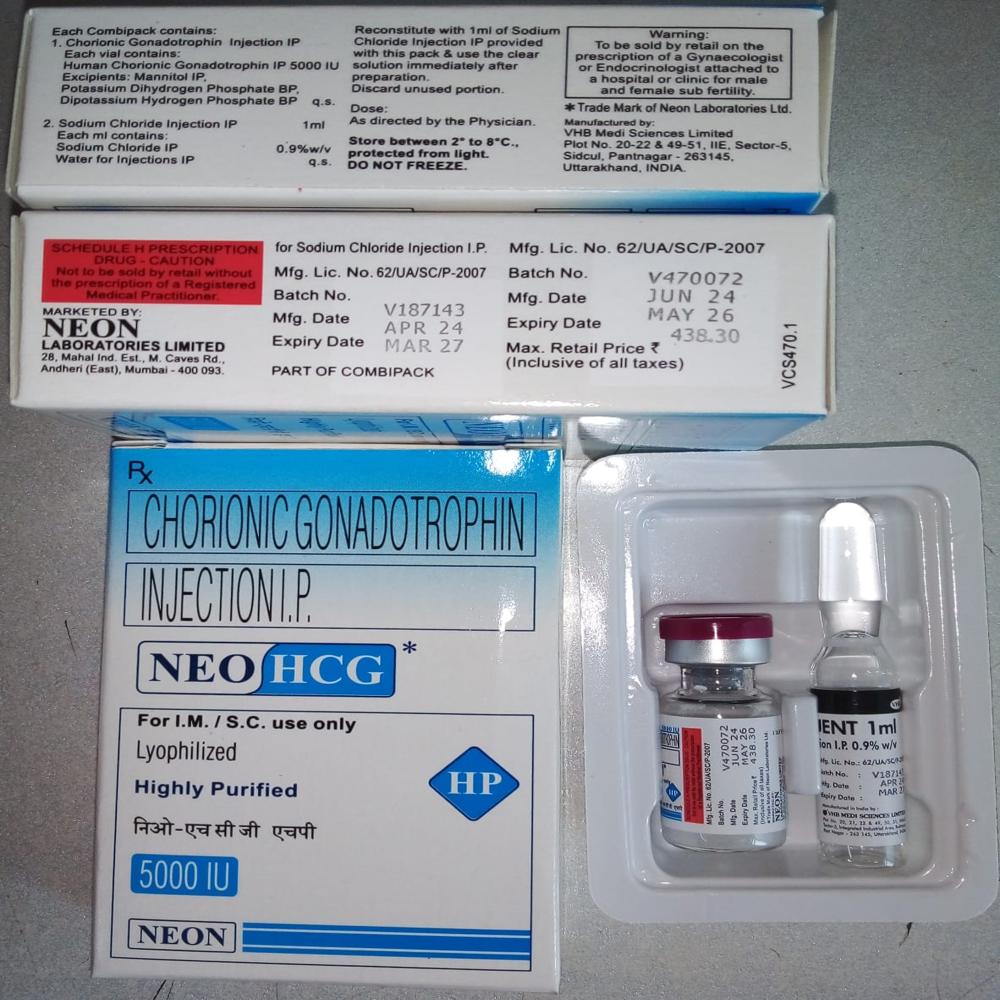
NEO HCG 5000IU
Price 190.0 INR / Box
Minimum Order Quantity : 50 Boxes
Dosage Guidelines : As per Instructions
Physical Form : Tablets
Drug Type : Injection
Storage Instructions : Cool & Dry Place
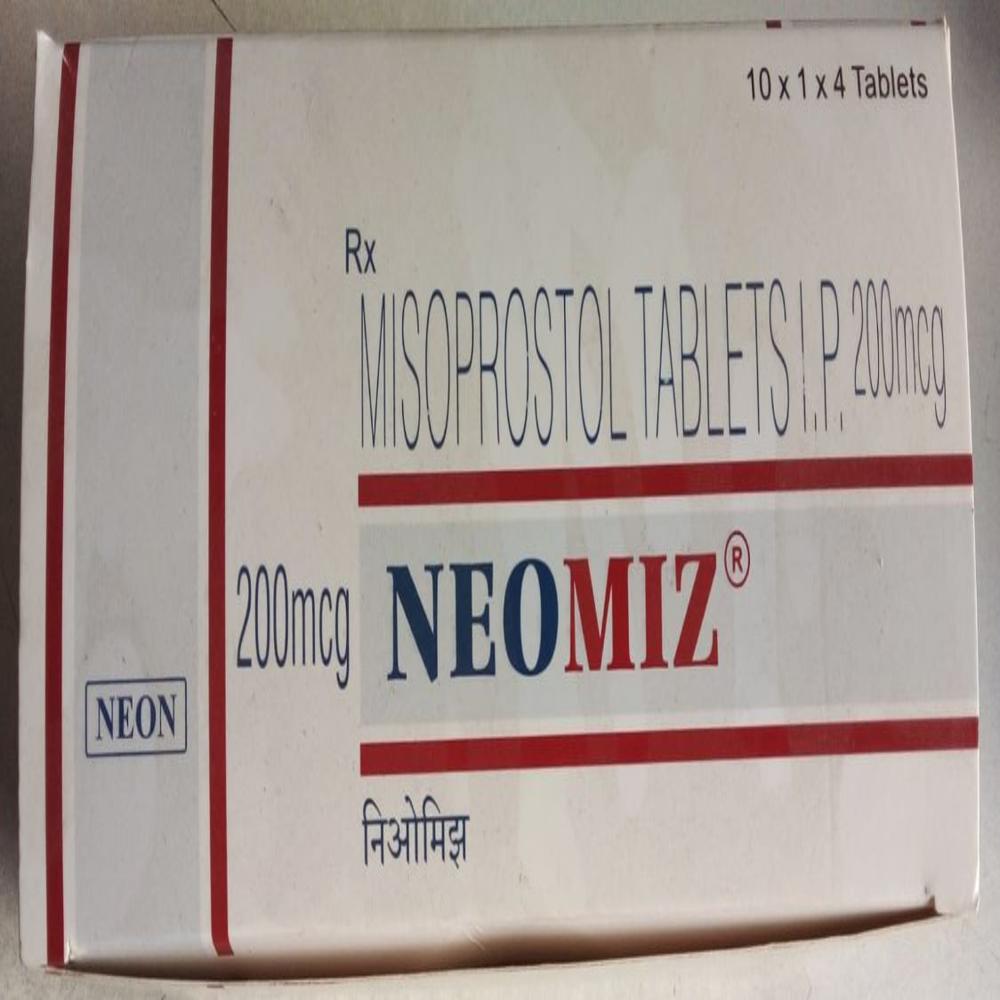
NEOMIZ 200MCG
Price 35.5 INR / Box
Minimum Order Quantity : 50 Boxes
Dosage Guidelines : As per Instructions
Physical Form : Liquid
Drug Type : Injection
Storage Instructions : Cool & Dry Place
VANKING 500MG
Price 77.0 INR / Unit
Minimum Order Quantity : 50 Units
Dosage Guidelines : As per Instructions
Physical Form : Liquid
Drug Type : Injection
Storage Instructions : Cool & Dry Place


 Send Inquiry
Send Inquiry



 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese