Working Hours
Mon - Sat 9:00 - 6:00SEVOTROY 50 ML
Price 1308.72 INR/ Vial
SEVOTROY 50 ML Specification
- Packaging Type
- Glass bottle
- Life Span
- 36 months from manufacture
- Indication
- Induction and maintenance of general anaesthesia
- Origin of Medicine
- India
- Dosage Form
- Liquid for inhalation
- Salt Composition
- Sevoflurane
- Pacakaging (Quantity Per Box)
- 1 bottle per box
- Brand Name
- SEVOTROY
- Drug Type
- Injection
- Ingredients
- SEVOFLURANE USP 50 ml
- Physical Form
- Liquid
- Function
- Inhalational Anaesthetic
- Recommended For
- Anaesthesia during surgical procedures
- Dosage
- As directed by the physician or anesthesiologist
- Dosage Guidelines
- To be administered only by inhalation under qualified medical supervision
- Suitable For
- Adults and Pediatric patients
- Quantity
- 50 ml
- Storage Instructions
- Store below 30C, protect from direct sunlight
- Country of Export
- India
- Expiry Date
- Printed on label
- Contraindications
- Known or suspected genetic susceptibility to malignant hyperthermia, hypersensitivity to sevoflurane or other halogenated agents
- Application Route
- Inhalation vapour
- Odor
- Mild, non-pungent odor
- Shelf Life After Opening
- Should be used within 28 days after opening when handled aseptically
- Regulatory Status
- Approved by Indian regulatory authorities for use in hospitals and operating rooms
- Appearance
- Clear, colorless, volatile liquid
- Flammability
- Non-flammable
- Boiling Point
- 58.5C
- Prescription Type
- Prescription Only (Rx)
- Batch Number
- Printed on outer carton and bottle
- Precautions
- Use with caution in patients with increased intracranial pressure or liver disorders
- Molecular Formula
- C4H3F7O
SEVOTROY 50 ML Trade Information
- Minimum Order Quantity
- 100 Vials
- Supply Ability
- 1000 Vials Per Month
- Delivery Time
- 7 Days
- Sample Available
- No
About SEVOTROY 50 ML
Specialized Use and Material Features
SEVOTROY 50 ML is a clear, volatile liquid designed exclusively for the induction and maintenance of general anaesthesia through inhalation. The commendable feature of this product is its non-flammable and mild odor profile, ensuring safety and comfort in clinical settings. Used as a prescription-only solution, it is recommended for both adults and pediatric patients, administered under strict medical supervision for optimal effectiveness and patient care.
Market Reach, Export Status, and Certifications
SEVOTROY 50 ML is distributed widely across India, with primary domestic and export markets including hospitals and large healthcare chains. Each shipped good adheres to Indian regulatory approvals and comes with verifiable batch and expiry information, reflecting transparency in the export market. The list price balances affordability and quality, ensuring it remains accessible to both local and international buyers seeking certified anaesthetic solutions.
FAQ's of SEVOTROY 50 ML:
Q: How should SEVOTROY 50 ML be administered for best results?
A: SEVOTROY 50 ML should only be administered by inhalation under the supervision of a qualified anesthesiologist or physician, following proper dosage guidelines.Q: What are the main indications for using SEVOTROY 50 ML?
A: SEVOTROY 50 ML is indicated for the induction and maintenance of general anaesthesia during surgical procedures in both adults and pediatric patients.Q: What precautions and contraindications should be considered before use?
A: It should be avoided in patients with known or suspected genetic susceptibility to malignant hyperthermia or hypersensitivity to sevoflurane or other halogenated agents. Caution is advised in patients with liver disorders or increased intracranial pressure.Q: Where can users find information on the batch number and expiry date of SEVOTROY 50 ML?
A: The batch number is printed on the outer carton and bottle, while the expiry date is printed on the label for easy reference and traceability.Q: When should SEVOTROY 50 ML be used after opening the bottle?
A: Once opened and handled aseptically, SEVOTROY 50 ML should be used within 28 days to ensure its effectiveness and safety.Q: What are the storage requirements for SEVOTROY 50 ML?
A: Store the product below 30C and protect it from direct sunlight to maintain its stability and effectiveness throughout its shelf life.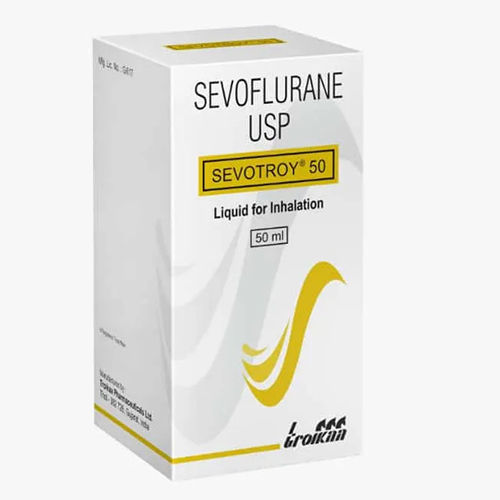

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in IV Fluids Category
TRUEPLAN 400MG
Price 600.0 INR / Unit
Minimum Order Quantity : 50 Units
Drug Type : Injection
Physical Form : Powder
Storage Instructions : ROOM TEMPRATURE
NORAD 2ML
Price 160 INR / Piece
Minimum Order Quantity : 20 Pieces
Drug Type : Injection
Physical Form : Liquid
Storage Instructions : Cool & Dry Place
Dosage Guidelines : As per Instructions
LIN IV 300ML
Price 77.28 INR / Bottle
Minimum Order Quantity : 100 Bottles
Drug Type : Injection
Physical Form : Liquid
Storage Instructions : Cool & Dry Place
Dosage Guidelines : As Per Suggestion
MUCYST 400MG/2ML
Price 19.67 INR / Box
Minimum Order Quantity : 50 Boxes
Drug Type : Injection
Physical Form : Liquid
Storage Instructions : Cool & Dry Place
Dosage Guidelines : As per Instructions


 Send Inquiry
Send Inquiry


 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese