Working Hours
Mon - Sat 9:00 - 6:00EFIPRES 30mg/ml
Price 29.76 INR/ Piece
EFIPRES 30mg/ml Specification
- Dosage Form
- Parenteral
- Packaging Type
- Ampoules in carton box
- Origin of Medicine
- India
- Life Span
- 24 months from manufacturing date
- Pacakaging (Quantity Per Box)
- 10 x 1 ml ampoules
- Salt Composition
- Adrenaline/Epinephrine 30mg/ml
- Brand Name
- EFIPRES
- Indication
- Anaphylaxis, Allergic emergencies, Cardiac arrest
- Drug Type
- Injection
- Ingredients
- Epinephrine Injection USP
- Physical Form
- Other
- Function
- Other
- Recommended For
- Acute anaphylaxis, Severe allergic reactions, Cardiac arrest management
- Dosage
- As prescribed by a physician
- Dosage Guidelines
- For intramuscular or intravenous use as directed by healthcare professional
- Quantity
- 10 Boxes
- Storage Instructions
- Store below 25C. Protect from light.
- Color
- Clear, colorless solution
- Route of Administration
- Intramuscular (IM), Intravenous (IV)
- Strength
- 30mg/ml
- Manufactured By
- Gufic Biosciences Ltd.
- Marketed By
- Gufic Biosciences Ltd.
- Sterility
- Sterile, pyrogen-free
- pH Range
- 2.2 - 5.0
- Controlled Substance
- No
- Regulatory Status
- Prescription only
EFIPRES 30mg/ml Trade Information
- Minimum Order Quantity
- 50 Pieces
- Supply Ability
- 1000 Pieces Per Month
- Delivery Time
- 7 Days
- Sample Available
- No
About EFIPRES 30mg/ml
Application & Site of Use
EFIPRES 30mg/ml is used as an emergency medication for acute anaphylactic episodes and severe allergic reactions. Application surfaces include intramuscular and intravenous sites, administered directly by healthcare professionals. The site of application is typically the large muscles (IM injection) or veins (IV injection) in both adults and children, ensuring quick, effective response during critical moments. It serves as a premier vasoconstrictor and is also essential in cardiac arrest management.
Export Market Reach & Dispatch Details
EFIPRES 30mg/ml has a remarkable export market presence, catering to international medical suppliers and hospitals. Packing & Dispatch are handled with utmost care-each purchase order is fulfilled using robust ampoule packaging in carton boxes, 10 x 1ml per box, ensuring product integrity during transit. The typical delivery time post-confirmation of purchase order is swift, guaranteeing on-time supply for emergency response units and medical facilities worldwide.
FAQ's of EFIPRES 30mg/ml:
Q: How is EFIPRES 30mg/ml administered in emergency situations?
A: EFIPRES 30mg/ml is administered via intramuscular or intravenous injection by a qualified healthcare professional, following your physician's instructions.Q: What are the primary uses of EFIPRES 30mg/ml?
A: EFIPRES is used for the emergency treatment of acute anaphylaxis, severe allergic reactions, and cardiac arrest, leveraging its rapid-acting vasoconstrictor properties.Q: When should EFIPRES 30mg/ml be used?
A: EFIPRES should be used immediately in life-threatening allergic emergencies, anaphylaxis, or cardiac arrest, as prescribed by a healthcare professional.Q: Where should EFIPRES 30mg/ml be stored for optimal effectiveness?
A: EFIPRES should be stored below 25C and protected from light to maintain its sterility and potency until its 24-month expiration.Q: What process ensures the sterility of EFIPRES 30mg/ml?
A: Each ampoule is manufactured under strict sterile, pyrogen-free conditions, ensuring high product safety and quality for medical emergencies.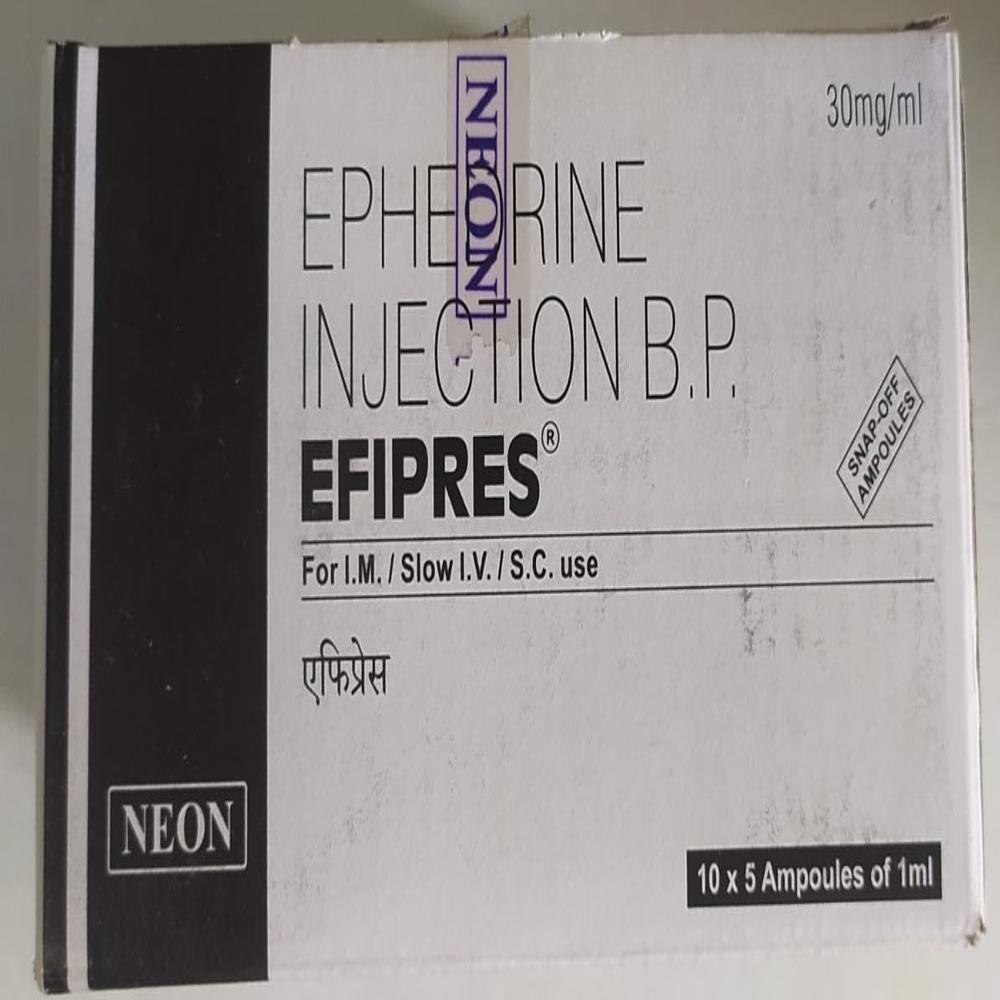

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in IV Fluids Category
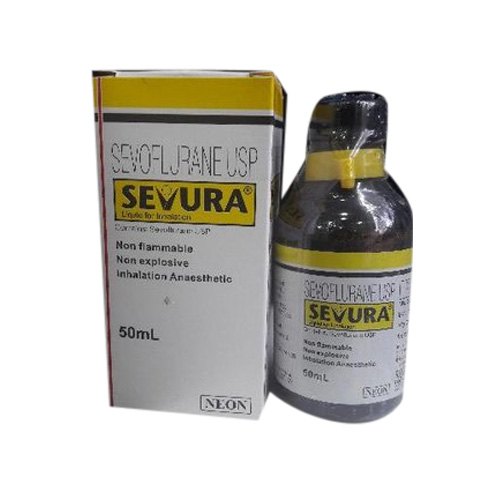
SEVURA 50ML
Price 1278.7 INR / Bottle
Minimum Order Quantity : 50 Bottles
Recommended For : Inhalational anaesthesia
Ingredients : Sevoflurane
Physical Form : Liquid
Drug Type : Other
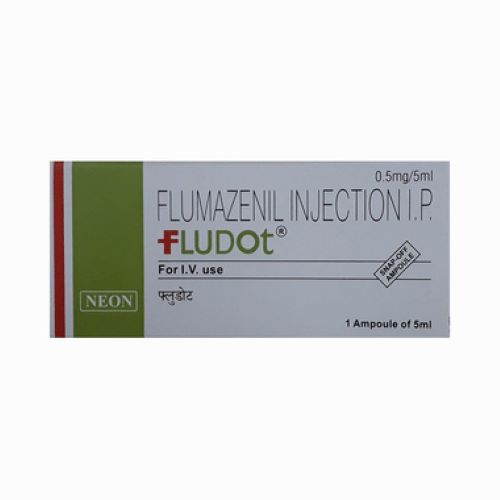
FLUDOT 0.5mg/5ml
Price 1048.0 INR / Unit
Minimum Order Quantity : 50 Units
Recommended For : Fungal Infections
Ingredients : FLUDOT 5ml Injection Flumazenil Injection Brand Name: FLUDOT 5ml Generic Name: Flumazenil Injection Dosage Form: Injection Pack Size: 1 Ampoule of 5 ml Manufacturer: Neon Laboratories Limited Product Description: FLUDOT 5ml Injection contains Flumazenil, a benzodiazepine receptor antagonist used to reverse the sedative effects of benzodiazepines. It is commonly used in anesthesia and critical care to promptly counteract excessive sedation or respiratory depression caused by benzodiazepine administration. Flumazenil works by competitively binding to GABA receptors, reversing the effects of benzodiazepines and restoring normal neurological function under controlled medical supervision. Common Uses: Reversal of benzodiazepineinduced sedation Management of benzodiazepine overdose Postoperative recovery from sedation ICU and critical care applications Key Features: Rapid onset of action Effective and safe benzodiazepine antagonist Hospitalgrade formulation Packaged in a convenient 5 ml ampoule Manufactured under strict GMP quality standards Note: To be administered only by qualified healthcare professionals in monitored clinical settings
Physical Form : Other
Drug Type : Injection
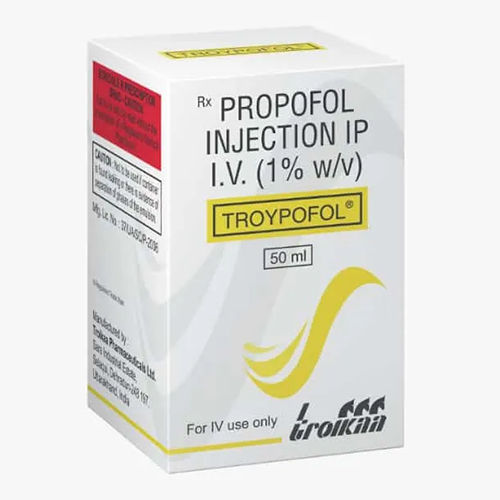
TROYPOFOL 50 ml
Price 168.0 INR
Minimum Order Quantity : 50
Recommended For : Patients undergoing surgical or diagnostic procedures
Ingredients : Propofol
Physical Form : Liquid
Drug Type : Other
TROYNEM 500
Price 208.0 INR
Minimum Order Quantity : 100
Recommended For : Patients with severe bacterial infections
Ingredients : Meropenem 500 mg
Physical Form : Other, Injection
Drug Type : Other, Antibiotic


 Send Inquiry
Send Inquiry


 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese